Vamorolone
New Drug Approvals
JULY 25, 2025
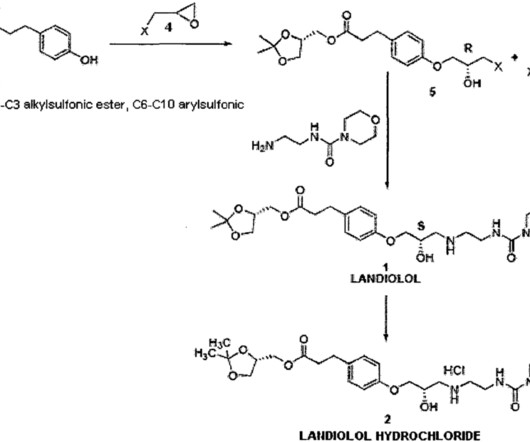
Syn EuropeanJournalofMedicinalChemistry265(2024)116124 Vamorolone (Agamree) On October 26, 2023, Vamorolone, developed jointly by Santhera Pharmaceuticals and ReveraGen BioPharma, has received FDA approval to treat DMD in patients aged 2 years and older [1]. 2025, 68, 2147−2182 Vamorolone (Agamree). Nagaraju, J.M. Damsker, J.M.










Let's personalize your content