UK approval for CBD pain treatment
Drug Discovery World
DECEMBER 2, 2022
The UK Medicines and Healthcare Products Regulatory Agency (MHRA) has approved a CBD/THC combination treatment under its Named Patient programme and other compassionate areas. Medlab’s NanaBis, to be used under NanaDol, a botanical version of NanaBis, has previously shown encouraging Phase I/II data for the treatment of cancer-induced bone pain and could be a potential alternative to opioids. .





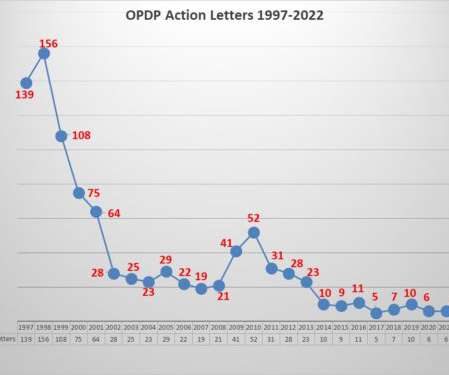

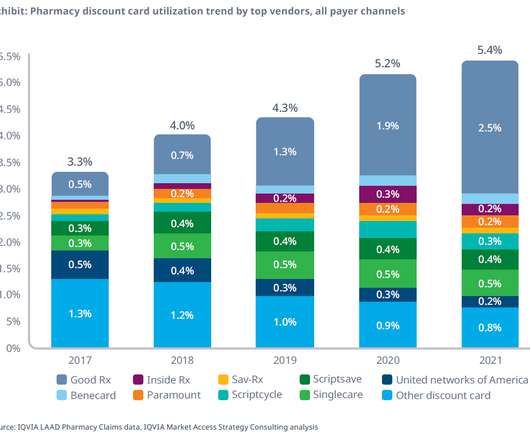




























Let's personalize your content