Biogen Launches Phase 3 Trial of Felzartamab for Primary Membranous Nephropathy
The Pharma Data
JULY 2, 2025
Biogen Launches Global Phase 3 PROMINENT Trial Evaluating Felzartamab in Primary Membranous Nephropathy Biogen Inc. The trial is expected to complete in 2029. This randomized, open-label study will compare felzartamab with tacrolimus, a commonly used immunosuppressant, in a cohort of approximately 180 patients worldwide.


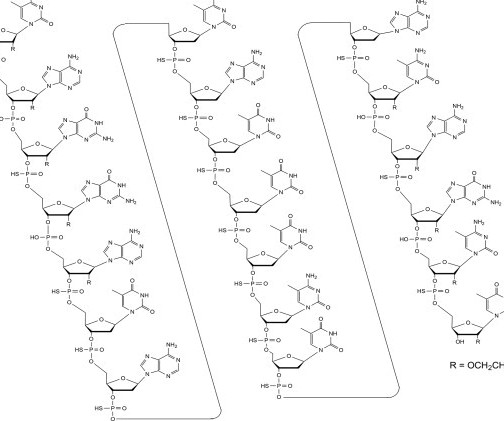
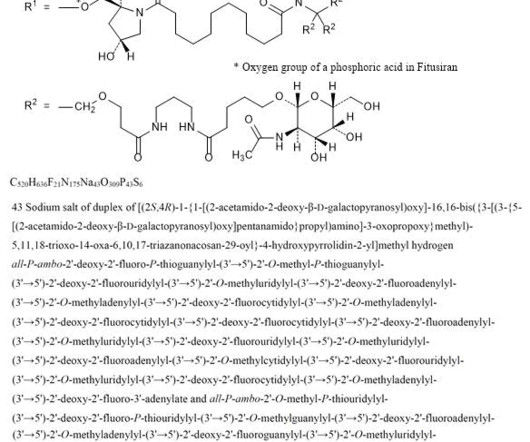
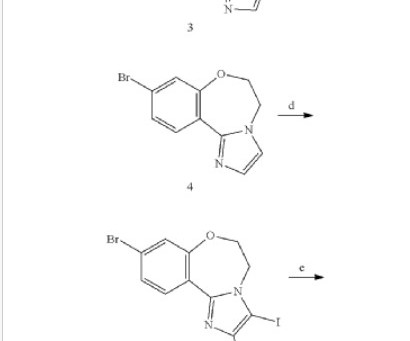
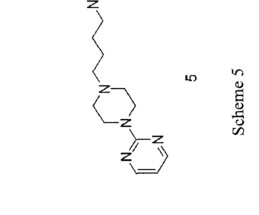



































Let's personalize your content