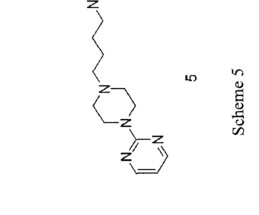
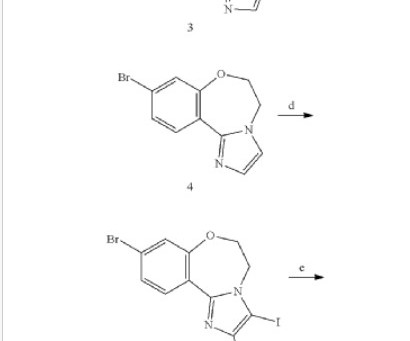
Gepirone
New Drug Approvals
JUNE 11, 2025
4] It was approved for the treatment of major depressive disorder in the United States in September 2023. [4] 4] This came after the drug had been rejected by the Food and Drug Administration (FDA) three times over two decades due to insufficient evidence of effectiveness. [5] The FDA rejected approval for gepirone in 2002 and 2004. [5]



















Let's personalize your content