FDA Approves CSL’s Andembry First Once-Monthly Prophylactic HAE Treatment Targeting Factor XIIa
The Pharma Data
JUNE 16, 2025
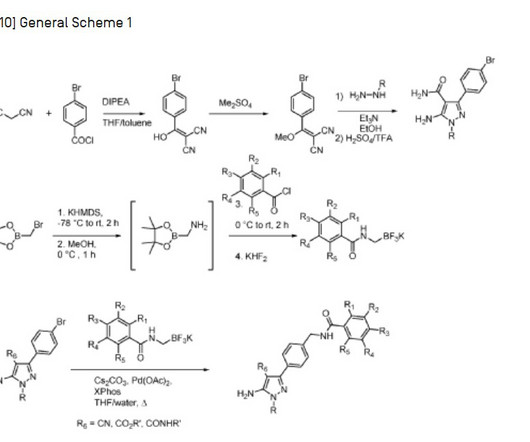
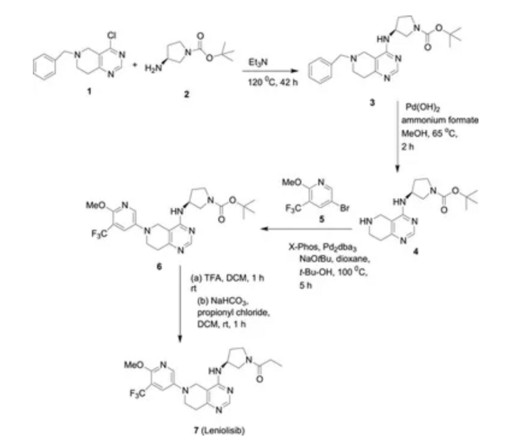
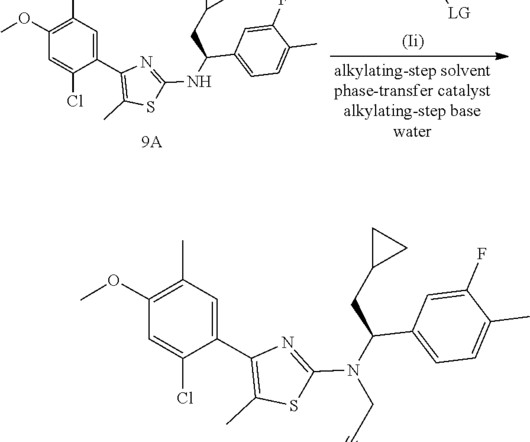
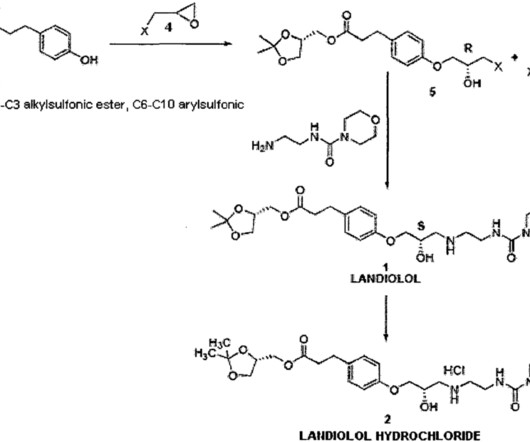
FDA Approves ANDEMBRY (garadacimab-gxii) The First Once-Monthly Prophylactic HAE Therapy Targeting Factor XIIa CSL a leading biotechnology company with a strong track record of developing innovative medicines for patients with rare and serious disorders, today announced that the U.S. when directly comparing ANDEMBRY to placebo.








































Let's personalize your content