Pfizer wins FDA approval for its $7B colitis drug
BioPharma Drive: Drug Pricing
OCTOBER 13, 2023
Acquired with the buyout of Arena Pharmaceuticals, Velsipity enters a competitive market for ulcerative colitis pills.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

BioPharma Drive: Drug Pricing
OCTOBER 13, 2023
Acquired with the buyout of Arena Pharmaceuticals, Velsipity enters a competitive market for ulcerative colitis pills.

Drugs.com
JANUARY 31, 2025
Food and Drug Administration (FDA) has approved Journavx, a new pain reliever without the risks of addiction or overdose linked to drugs like Vicodin and OxyContin.The new pill, developed by Vertex Pharmaceuticals.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

SCIENMAG: Medicine & Health
OCTOBER 31, 2023
— A new drug developed by professors from the School of Pharmacy and Pharmaceutical Sciences at Binghamton University has received Food and Drug Administration (FDA) approval for the treatment of patients with Duchenne muscular dystrophy (DMD), a common genetic disease that mostly affects young boys. BINGHAMTON, N.Y.

Drug Patent Watch
JULY 24, 2024
In the high-stakes world of pharmaceuticals, generic drugs have become the unsung heroes of healthcare accessibility. These cost-effective alternatives to […] Source
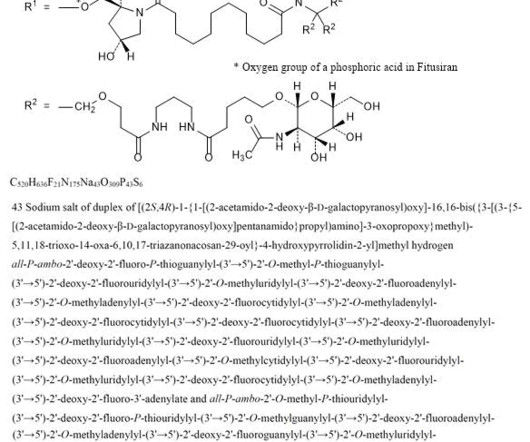
New Drug Approvals
APRIL 11, 2025
2] Fitusiran was approved for medical use in the United States in March 2025. [2] 2] The fixed dose of fitusiran is not approved because it led to excessive clotting in some participants. [2] 2] The US Food and Drug Administration (FDA) granted the application for fitusiran orphan drug and fast track designations. 26 March 2025.

The Pharma Data
JUNE 9, 2025
Xywav: A Low-Sodium Alternative with FDA Approval Xywav is a uniquely formulated, low-sodium oxybate therapy, and remains the only product of its kind approved by the U.S. It is also approved for adult patients with idiopathic hypersomnia (IH). Source link
New Drug Approvals
JUNE 11, 2025
2] Gepirone was synthesized by Bristol-Myers Squibb in 1986 and was developed and marketed by Fabre-Kramer Pharmaceuticals. [4] 4] It was approved for the treatment of major depressive disorder in the United States in September 2023. [4] 12] However, in March 2016, the FDA reversed its decision and gave gepirone ER a positive review.

The Pharma Data
AUGUST 27, 2020
The test is FDA-approved to report short variants in 311 genes including rearrangements and copy number losses in BRCA1 and BRCA2 genes. The results are delivered in an integrated report that identifies alterations matched to FDA-approved therapies.

Metabolite Tales Blog
JANUARY 26, 2023
Metabolism of 2022 FDA approved small molecule drugs – Part 1 Does CYP3A4 still rule? By Julia Shanu-Wilson It won’t come as much surprise to learn that of the 17 small molecules* approved by the FDA in 2022, CYP3A4 was the major player in drug metabolism. References Iversen et al., Front Pharmacol.,

The Pharma Data
NOVEMBER 2, 2020
FDA Approves Bronchitol (mannitol) Inhalation Powder to Improve Pulmonary Function in Adult Patients with Cystic Fibrosis. Food and Drug Administration (FDA) approval of Bronchitol (mannitol) inhalation powder. Food and Drug Administration (FDA) approval of Bronchitol (mannitol) inhalation powder.

Drug Patent Watch
APRIL 8, 2025
Patient Safety in the Era of Generic Drugs: A Call to Action As the pharmaceutical industry continues to evolve, one thing remains constant: the need for patient safety. Check the FDA approval status : Make sure the generic medication has been approved by the FDA and meets all necessary safety and efficacy standards.

Agency IQ
JANUARY 12, 2024
After a years-long review process, the FDA today announced its approval of Florida’s proposal to import certain prescription drug products from Canada. Fill out the form to read the full article.

The Pharma Data
DECEMBER 16, 2020
17, 2020 /PRNewswire/ — CNS Pharmaceuticals, Inc. We will now rapidly move to initiate our Phase 2 trial of Berubicin for adults with GBM and expect to begin enrolling patients in the first quarter of next year,” commented John Climaco , CEO of CNS Pharmaceuticals. About CNS Pharmaceuticals, Inc.

The Pharma Data
NOVEMBER 27, 2020
FDA Approves Danyelza (naxitamab-gqgk) for the Treatment of Neuroblastoma. It’s very exciting to see this treatment go from being an experimental therapy used at my daughter’s bedside to now being FDA approved. Related Articles: Danyelza (naxitamab-gqgk) FDA Approval History. NEW YORK, Nov. Source link.

The Pharma Data
NOVEMBER 27, 2020
FDA Approves Imcivree (setmelanotide) for Chronic Weight Management in Patients with Obesity Due to POMC, PCSK1 or LEPR Deficiency. 27, 2020 (GLOBE NEWSWIRE) — Rhythm Pharmaceuticals, Inc. With this approval, Imcivree becomes the first-ever FDA approved therapy for these rare genetic diseases of obesity.
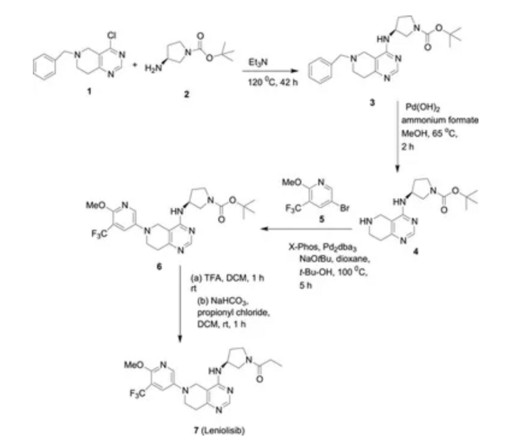
New Drug Approvals
MAY 15, 2025
5] Leniolisib was approved for medical use in the United States in March 2023. [5] 5] [7] [8] It is the first approved medication for the treatment of activated PI3K delta syndrome. [5] 5] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication. [9] “Leniolisib: First Approval” Drugs.

Pharmaceutical Development Group
AUGUST 9, 2021
Formulation chemistry is the systematic and step-by-step approach to pharmaceutical development. Nowadays, as the usage of medicines is increasing, pharmaceutical companies are more eager to bring manufacturing a new look in quality and performance. There are different tests to assess the quality of a pharmaceutical product.

The Pharma Data
DECEMBER 15, 2020
FDA Approves Klisyri (tirbanibulin) for the Treatment of Actinic Keratosis on the Face or Scalp. Food and Drug Administration (FDA) has approved Klisyri (tirbanibulin) for the topical treatment of actinic keratosis (AK) on the face or scalp. The FDA approval of Klisyri is a significant milestone for Athenex.

FDA Law Blog: Biosimilars
OCTOBER 1, 2024
Karst — If you monitor Regulations.gov dockets and litigation dockets on PACER like we do, then you know that one company name—more than any other over the past several years—pops up: Vanda Pharmaceuticals, Inc. VANDA PHARMACEUTICALS, INC. VANDA PHARMACEUTICALS, INC. 24-270 VANDA PHARMACEUTICALS, INC. 23-5200 (D.C.

The Pharma Data
MAY 27, 2022
Novartis today announced the US Food and Drug Administration (FDA) has granted accelerated approval for Kymriah ® (tisagenlecleucel) for the treatment of adult patients with relapsed or refractory (r/r) follicular lymphoma (FL) after two or more lines of systemic therapy.

Advarra
JULY 27, 2022
As a pharmaceutical product makes its way through the lifecycle, there are often Food and Drug Administration (FDA) guidelines organizations should pay particular attention to. If your go-to-market strategy requires FDA approval, it may also require a prior approval supplement. Go-to-market Strategy.

The Pharma Data
DECEMBER 1, 2020
VICTORIA, British Columbia–( BUSINESS WIRE )– Aurinia Pharmaceuticals Inc. Aurinia Pharmaceuticals is a late-stage clinical biopharmaceutical company focused on developing and commercializing therapies to treat targeted patient populations that are impacted by serious diseases with a high unmet medical need. ABOUT AURINIA.

Drug Target Review
JANUARY 8, 2024
Jerry has over 30 years of experience in the biopharmaceutical industry and has been involved in the discovery, clinical development, and global commercialisation of more than a dozen FDA-approved drugs with multiple successful exits. a commercial stage pharmaceutical company. acquired by Teva Pharmaceuticals in 2014.

Drug Patent Watch
APRIL 29, 2025
"Market Access: The Hidden Hurdle in Generic Drug Commercialization As a seasoned pharmaceutical professional, I've seen it time and time again: a generic drug finally receives FDA approval, only to stall in the market due to a seemingly insurmountable obstacle - market access.

The Pharma Data
OCTOBER 21, 2020
Food and Drug Administration (FDA) to market Ephedrine Sulfate Injection in a ready-to-use 50mg/10 ml single use vial presentation. Under an exclusive licensing agreement with Endo International’s (NASDAQ: ENDP) subsidiary, Endo Ventures Limited, Par Pharmaceuticals’ Sterile Products division will launch and distribute the product.

The Pharma Data
JANUARY 7, 2021
VICTORIA, British Columbia–( BUSINESS WIRE )– Aurinia Pharmaceuticals Inc. Aurinia Pharmaceuticals is a late-stage clinical biopharmaceutical company focused on developing and commercializing therapies to treat targeted patient populations that are impacted by serious diseases with a high unmet medical need. ABOUT AURINIA.

The Pharma Data
APRIL 19, 2022
Food and Drug Administration (FDA) has approved commercial production at the company’s new CAR T-cell therapy manufacturing facility in Frederick, Maryland. The site will produce Kite’s FDA approved CAR T-cell therapy used to treat blood cancer.

The Pharma Data
JUNE 11, 2025
Ionis Begins Pivotal Phase 3 REVEAL Study of ION582 in Angelman Syndrome, Dosing First Patient in Global Trial Ionis Pharmaceuticals , Inc. Ionis Pharmaceuticals has a well-established track record in the development of antisense therapies for neurological disorders.
New Drug Approvals
APRIL 25, 2025
3] Inavolisib was approved for medical use in the United States in October 2024. [3] 19] Society and culture Legal status In October 2024, the US Food and Drug Administration (FDA) approved inavolisib for the treatment of PIK3CA -mutant breast cancer based on the results from the INAVO120 trial. [3] 3 November 2006. PMC 8706051.

The Pharma Data
AUGUST 31, 2020
Food and Drug Administration (FDA) approval for the cobas® HIV-1/HIV-2 Qualitative Test for use on the fully automated cobas® 6800/8800 Systems in the U.S. About Roche Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. Learn more now: www.cobas68008800.com.
The Pharma Data
JULY 29, 2021
The FDA released new materials for health care providers to enhance understanding about biosimilar and interchangeable biosimilar products, including a fact sheet about interchangeable biosimilar products. The FDA granted approval of Semglee (insulin glargine-yfgn) to Mylan Pharmaceuticals Inc. Source link: [link].

DrugBank
DECEMBER 18, 2024
A new shift is occurring in the pharmaceutical industry, leading to a rapidly expanding field known as digital therapeutics (DTx). DTx interventions include sensor-equipped wearable devices, remote patient monitoring tools, and virtual reality platforms integrated with conventional pharmaceutical treatments.
Drug Target Review
JUNE 5, 2025
Currently, three FDA-approved disease-modifying drug therapies are available: hydroxyurea, crizanlizumab and L-glutamine, though each has limitations that affect patient compliance. The use of FDA-approved medications for preventing vaso-occlusive events in sickle cell disease. JAMA Netw Open. 2023 Nov 1;6(11):e2344546.
The Pharma Data
DECEMBER 3, 2020
BioCryst Pharmaceuticals, Inc. Food and Drug Administration (FDA) has approved ORLADEYO (berotralstat) for prophylaxis to prevent attacks of hereditary angioedema (HAE) in adults and young patients 12 years and older. announced on Thursday that the U.S.
Drug Target Review
MARCH 12, 2024
Scientists from the Skaggs School of Pharmacy and Pharmaceutical Sciences at the University of California San Diego have discovered thousands of bile acids. The study’s findings offer new insights into the biochemical language microbes use to influence distant organ systems, which could revolutionise the way researchers approach disease.
New Drug Approvals
APRIL 18, 2025
2] Crinecerfont was approved for medical use in the United States in December 2024. [2] 2] [3] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication. [4] 2] The FDA granted the approval of Crenessity to Neurocrine Biosciences, Inc. [2] Food and Drug Administration (FDA) (Press release).
New Drug Approvals
APRIL 2, 2025
1] [2] It was developed by Vertex Pharmaceuticals , [5] and was approved for medical use in the United States in January 2025. [2] 2] [6] Suzetrigine is the first medication to be approved by the US Food and Drug Administration (FDA) in this new class of pain management medicines. [2] Food and Drug Administration (FDA).

Pharmaceutical Development Group
DECEMBER 19, 2021
Start Up and Generic Pharmaceutical Drug and Biologic Companies have high quality, affordable products that improve the quality of life for their patients. In a FDA Pre-Submission that leads to FDA Approval, more does not equal better and more does not equal relevance to a specific Pharmaceutical Drug or Biologic Product.

Pharmaceutical Development Group
JANUARY 7, 2022
Start Up and Generic Pharmaceutical Drug and Biologic Companies have high quality, affordable products and biosimilars that improve the quality of life for their patients. Dr. Spanogle has taken scores of Regulated Life Science Products from discovery to approval and launch; and audited, identified, and remediated QMS deficiencies.

FDA Law Blog: Biosimilars
JUNE 27, 2023
Amongst his accomplishments, Law360 considered the role James has played in leveraging little-used pathways to FDA approval for often first-ever drugs to treat rare diseases (e.g., James was only one of five life sciences attorneys selected and the only food and drug lawyer to make the list.
The Pharma Data
NOVEMBER 2, 2020
GW Pharmaceuticals hopes to bring its cannabis-based treatment for multiple sclerosis spasticity to the United States. Sativex is approved for use in parts of Europe for this indication. Justin Gover, chief executive officer of GW Pharmaceuticals, proclaimed his excitement about launching the Phase III study in the United States.
The Pharma Data
JULY 8, 2021
is a leading global pharmaceutical company headquartered in Japan. We routinely post information that may be important to investors on our website at www.biogen.com. Follow us on social media – Twitter , LinkedIn , Facebook , YouTube. About Eisai Co.,

The Pharma Data
JANUARY 6, 2021
SK Chemicals’ life sciences business is dominated by pharmaceuticals and biologics and is aimed at providing comprehensive healthcare solutions that cover patient care from diagnosis to treatment and prevention.

The Pharma Data
DECEMBER 16, 2020
–( BUSINESS WIRE )– ANI Pharmaceuticals, Inc. , (“ANI” or the “Company”) (Nasdaq: ANIP) today announced U.S. Food and Drug Administration (FDA) approval and the launch of Aminocaproic Acid Tablets USP, 500mg. ANI Pharmaceuticals, Inc. BAUDETTE, Minn.–( The current annual U.S. earlier this year.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content